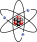
Rutherford theory says that in the centre of an atom is a small positively charged nucleus, which is surrounded by the negatively charged electrons. Content: Bohr Vs Rutherford’s Atomic Models With reference to these theories, in this article, we will be highlighting the difference between the two, with a brief explanation of their work. 
Moving onto the 18th century when Dalton’s proposed the atomic theory and then passing in the 20th century with the discovery of subatomic particles, the plum pudding model, Rutherford model, Bohr model, and quantum theory, the journey was full of discoveries and information.īut the two milestones along the whole path is the Bohr atomic theory, which was the modified version of the Rutherford atomic model and so sometimes named as Rutherford-Bohr atomic model. So, it can be said that the Atomic theory has crossed the thousands of years, when it begins in the 5th century B.C., with Democritus theory “ matter consists of the indivisible, indestructible units that interact with each other“. Though Atomic theory is the concept that originated in ancient India and Greece. The word ‘atoms’ is derived from the Greek word ‘ atomos‘, meaning ‘ indivisible‘. The Atomic theory is the scientific explanation of the properties of atoms and matter that combines factors of physics, chemistry, and mathematics. We all are aware of the modern theory which says that “matter is made up of tiny particles known as atoms which in turn are made up of subatomic particles”. 
Ernest Rutherford said that an atom consists of the whole mass that is concentrated into the center.įrom the above statement, we can say that both the models of the atom differ in the way they explained the movements of electrons around the nucleus and their energy levels. Niels Bohr postulates the atomic model which states that electrons move in specific circular orbits around the nucleus with quantized kinetic and potential energies.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
